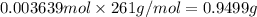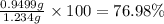The concentration of barium ions in any solution can also be determined via gravimetric analysis. an impure sample of barium nitrate with a mass of 1.234 g, is completely dissolved in water and the resulting solution is reacted with an excess of aqueous sodium sulfate. a precipitate forms, and after filtering and drying, it was found to have a mass of 0.848 g.
a) what is the relevance of adding eccess sodium sulfate?
b) calculate the % of barium nitrate in the original 1.234 g sample.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:00, levelebeasley1
Which theory was contradicted by experiments with the photoelectric effect? light spreads out after it passes through a small opening. as soon as light strikes metal, electrons will be ejected. visible light, regardless of color, will cause the ejection of electrons when striking metal. the kinetic energy of ejected electrons depends on the frequency of light that strikes the metal.
Answers: 2


Chemistry, 22.06.2019 22:30, teagan56
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1

Chemistry, 22.06.2019 22:30, StupidFatChipmunk
What must be in balance for temperatures to remain constant?
Answers: 1
Do you know the correct answer?
The concentration of barium ions in any solution can also be determined via gravimetric analysis. an...
Questions in other subjects:



History, 03.08.2019 19:00

History, 03.08.2019 19:00

Mathematics, 03.08.2019 19:00




Computers and Technology, 03.08.2019 19:00


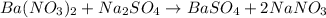
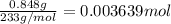
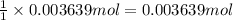 of barium nitrate.
of barium nitrate.