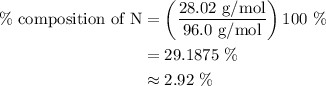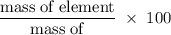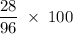Chemistry, 15.10.2019 02:30, lexhorton2002
What is the percent composition by mass of nitrogen in (nh4)2co3 (gram-formula mass = 96.0 g/mol)?
(1) 14.6%
(2) 29.2%
(3) 58.4%
(4) 87.5%

Answers: 1
Other questions on the subject: Chemistry



Do you know the correct answer?
What is the percent composition by mass of nitrogen in (nh4)2co3 (gram-formula mass = 96.0 g/mol)?...
Questions in other subjects:

Mathematics, 16.10.2019 12:00







Mathematics, 16.10.2019 12:00

Biology, 16.10.2019 12:00


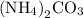 is
is 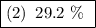 .
.
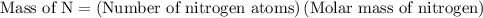 …… (1)
…… (1)
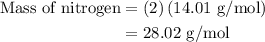
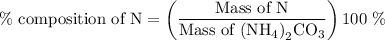 …… (2)
…… (2)