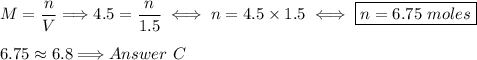How many moles of glucose (c6h12o6) are in 1.5 liters of a 4.5 m c6h12o6 solution?
4.5...

Chemistry, 04.02.2020 18:56, ghari112345
How many moles of glucose (c6h12o6) are in 1.5 liters of a 4.5 m c6h12o6 solution?
4.5 moles
6.0 moles
6.8 moles
3.0 moles

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:10, CauseWhyNot6235
What can be added to the examples section of each circle? endothermic: ice melting into water, and a heat pack becoming warm exothermic: a glow stick glowing, and fireworks exploding endothermic: ice melting into water, and an instant ice pack turning cold exothermic: fireworks exploding, and gasoline burning endothermic: a glow stick glowing, and a heat pack becoming warm exothermic: an instant ice pack turning cold, and ice melting into water endothermic: gasoline burning, and an instant ice pack turning cold exothermic: ice melting into water, and an instant ice pack turning cold
Answers: 1


Chemistry, 22.06.2019 13:00, rome58
Lab reagent, hypothesis test. a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl. these six measurements are assumed to be an srs of all possible measurements from solution. they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution. carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1
Do you know the correct answer?
Questions in other subjects:



Mathematics, 13.10.2019 20:30





History, 13.10.2019 20:30



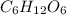 , n is the number of moles and V is the volume of the solution:
, n is the number of moles and V is the volume of the solution: