Chemistry, 19.01.2020 01:31, clarissajimenez27
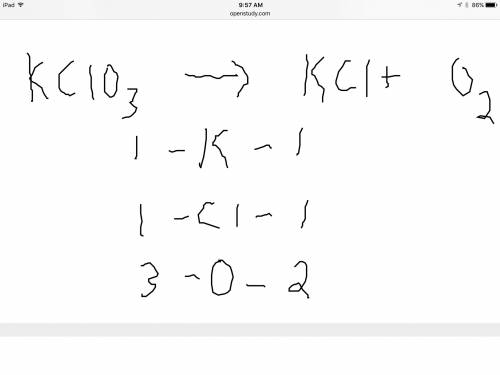
Balance the following equation. then determine the ratio for the products kcl and o2 generated during the decomposition of potassium chlorate. kclo3 kcl + o2

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, Luzperez09
Initially, the balloon had 3.0 liters of gas at a pressure of 400 kpa and was at a temperature of 294 k. if the balloon is cooled to 277 k and its volume decreased to 1 l, what will the new pressure in the balloon be?
Answers: 1


Chemistry, 22.06.2019 03:30, ilizzy1224
Adrop of acetone (nail polish remover) has a mass of 35 mg and a density of 0.788 g/cm3. what is its volume in cubic centimeters?
Answers: 3
Do you know the correct answer?
Balance the following equation. then determine the ratio for the products kcl and o2 generated durin...
Questions in other subjects:

Mathematics, 18.03.2021 19:20

History, 18.03.2021 19:20

History, 18.03.2021 19:20

English, 18.03.2021 19:20

English, 18.03.2021 19:20


Mathematics, 18.03.2021 19:20

English, 18.03.2021 19:20

Social Studies, 18.03.2021 19:20