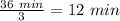Chemistry, 30.01.2020 08:46, mariahcid904
A24-gram sample of a radioactive nuclide decayed to 3.0 grams of the nuclide in 36 minutes. how much of the original nuclide sample remained after the first 12 minutes?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 03:30, ilizzy1224
Adrop of acetone (nail polish remover) has a mass of 35 mg and a density of 0.788 g/cm3. what is its volume in cubic centimeters?
Answers: 3


Chemistry, 22.06.2019 18:00, kyllow5644
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3
Do you know the correct answer?
A24-gram sample of a radioactive nuclide decayed to 3.0 grams of the nuclide in 36 minutes. how much...
Questions in other subjects:

History, 24.09.2019 08:50



History, 24.09.2019 08:50

Mathematics, 24.09.2019 08:50



Mathematics, 24.09.2019 08:50

Spanish, 24.09.2019 08:50

Business, 24.09.2019 08:50


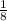 of its original size, so it underwent
of its original size, so it underwent 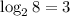 half-lives.
half-lives.