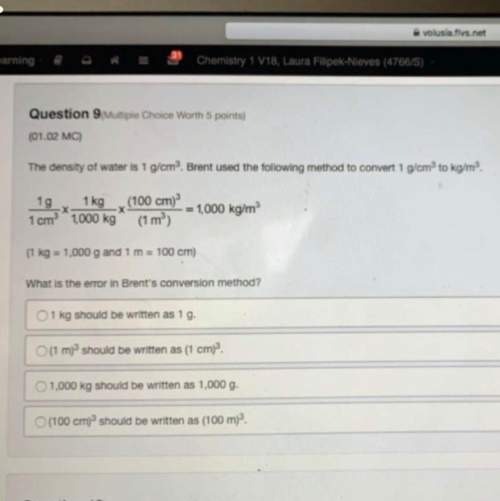
What volume of h2o(g) is produced when 8.00 mol of c2h4(g) reacts at stp?
c2h4 + 3o2 &g...

Chemistry, 09.10.2019 10:30, alexis9263
What volume of h2o(g) is produced when 8.00 mol of c2h4(g) reacts at stp?
c2h4 + 3o2 > > > > > 2co2 + 2h2o

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, rebeccacruzz2017
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 22.06.2019 12:40, valenzueladomipay09u
How does concentration affect reaction rate
Answers: 2

Do you know the correct answer?
Questions in other subjects:

Physics, 16.07.2019 08:30


Mathematics, 16.07.2019 08:30


Mathematics, 16.07.2019 08:30

SAT, 16.07.2019 08:30


English, 16.07.2019 08:30

Mathematics, 16.07.2019 08:30

History, 16.07.2019 08:30