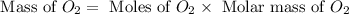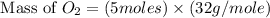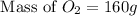Answers: 2
Other questions on the subject: Chemistry


Chemistry, 21.06.2019 23:00, carter1809
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2

Chemistry, 22.06.2019 12:50, khorasanpublic
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 16:00, winnie45
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2
Do you know the correct answer?
How many grams of o2 are in 5.0 moles of the oxygen gas?...
Questions in other subjects:


History, 27.09.2019 05:10

History, 27.09.2019 05:10


Mathematics, 27.09.2019 05:10



History, 27.09.2019 05:10

Mathematics, 27.09.2019 05:10

Physics, 27.09.2019 05:10


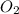 = 5 moles
= 5 moles