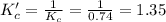Chemistry, 19.04.2021 18:20, jeremiaht7
At some temperature for the equilibrium PX3(g) + X2(g) # PX5(g) the equilibrium constant is 0.74. At the same temperature the equilibrium constant for PX5(9) + PX3(g) X2(g) is

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, logan12345677885675
What are the primary responsibilities of a chemical engineer involved in "r& d"? develop large scale manufacturing operations discover new products and processes training of new chemists determine products needed by consumers
Answers: 2

Chemistry, 23.06.2019 00:00, scottykinkade7860
Mercury turns to a vapor at 629.88 k. how much heat is lost when 75.0 g of mercury vapor at 650 k condenses to a liquid at 297 k?
Answers: 1

Chemistry, 23.06.2019 04:00, winterblanco
If you are told to get 100 ml of stock solution to use to prepare smaller size sample for an experiment, which piece of glassware would you use?
Answers: 1
Do you know the correct answer?
At some temperature for the equilibrium PX3(g) + X2(g) # PX5(g) the equilibrium constant is 0.74. At...
Questions in other subjects:

Chemistry, 13.02.2022 01:00

Mathematics, 13.02.2022 01:00

Mathematics, 13.02.2022 01:00

Mathematics, 13.02.2022 01:00



English, 13.02.2022 01:00

English, 13.02.2022 01:00

English, 13.02.2022 01:00


 is 1.35
is 1.35

![K_c=\frac{[PX_5]^1}{[PX_3]^1[X_2]^1}](/tpl/images/1268/5086/e23fb.png)
![0.74=\frac{[PX_5]^1}{[PX_3]^1[X_2]^1}](/tpl/images/1268/5086/ef131.png)
 is written as:
is written as:
![K_c'=\frac{[PX_3]^1[X_2]^1}{[PX_5]^1}](/tpl/images/1268/5086/99777.png)