
Chemistry, 16.04.2021 02:10, bradydodson47
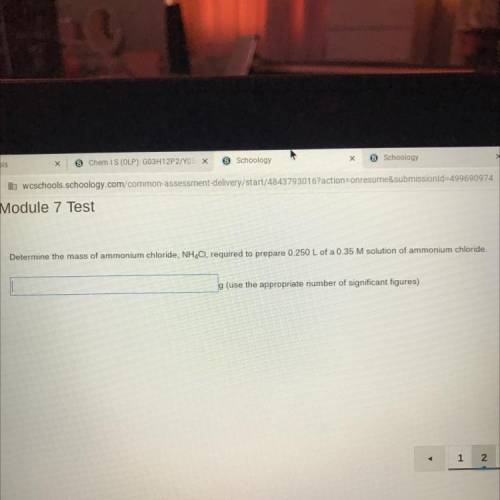
Determine the mass of ammonium chloride, NH4CI, required to prepare 0.250 L of a 0.35 M solution of ammonium chloride.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:20, missayers172
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

Chemistry, 22.06.2019 13:00, wbrandi118
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3

Do you know the correct answer?
Determine the mass of ammonium chloride, NH4CI, required to prepare 0.250 L of a 0.35 M solution of...
Questions in other subjects:















