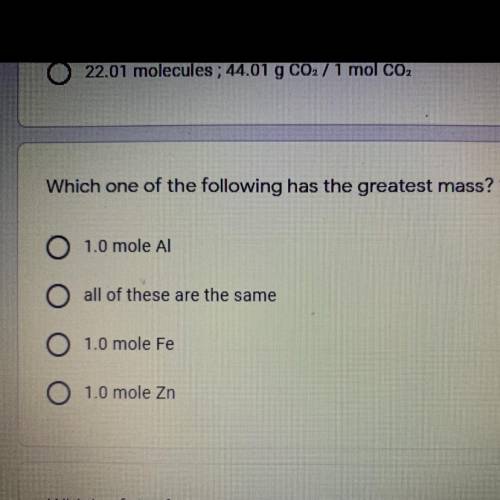
Which one of the following has the greatest mass? Help, please!
...

Chemistry, 14.04.2021 20:50, jeffyisdrunk
Which one of the following has the greatest mass? Help, please!

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, pinapunapula
Which explanation is true about what happens to a ray of light when it strikes a mirror? a. a light ray is transmitted toward a mirror at a certain angle. the light ray is then reflected by the mirror at an equal angle but in the opposite direction of the transmitted ray. b. an incident ray strikes a mirror at an angle with a line perpendicular to the mirror. the light ray is then reflected at an angle equal to the angle of incidence but on the opposite side of the perpendicular line. c. a reflecting ray strikes a mirror at an angle with a line perpendicular to the mirror. the light ray is then refracted at an angle equal to the angle of the reflecting ray and on the same side of the perpendicular line. d. an incident ray strikes a mirror at an angle with a line parallel to the mirror. the light ray is then transmitted at an angle equal to the angle of incidence but on the opposite side of the parallel line. you so much! : -d take the time to try and answer correctly.
Answers: 3

Chemistry, 22.06.2019 09:10, chloeholt123
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Do you know the correct answer?
Questions in other subjects:

Mathematics, 02.08.2019 10:30

Mathematics, 02.08.2019 10:30






Mathematics, 02.08.2019 10:30


Mathematics, 02.08.2019 10:30






