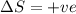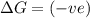Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, gatorr2010
An alcohol thermometer makes use of alcohol's changing in order to measure temperature. as the temperature goes up, the alcohol contained in the thermometer increases in volume, filling more of the thermometer's tube.
Answers: 3

Chemistry, 22.06.2019 05:30, NorbxrtThaG
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 11:10, hannah2757
Which of the following shapes would represent a molecule with two bonded atoms and 3 lone pairs on only one of them , trigonal planar , bent , trigonal pyramidal , linear
Answers: 1
Do you know the correct answer?
What does a process require to be spontaneous at all temperatures? answer a catalyst and lower acti...
Questions in other subjects:

Arts, 11.02.2021 01:00


Chemistry, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00

Health, 11.02.2021 01:00


Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00



 = free energy change
= free energy change = enthalpy change
= enthalpy change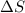 = entropy change
= entropy change , reaction is spontaneous
, reaction is spontaneous