
Chemistry, 14.04.2021 03:00, brittneyrost3722
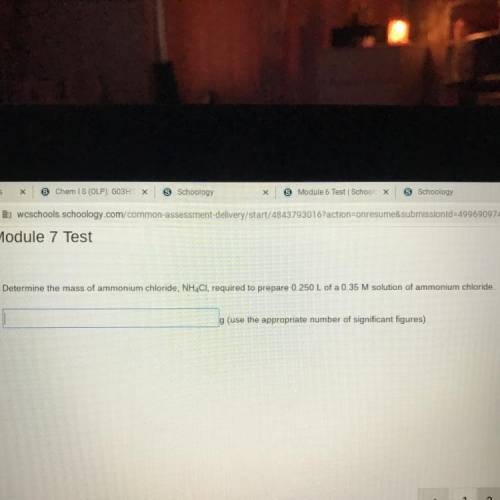
Determine the mass of ammonium chloride, NH4Cl, required to prepare 0.250 L of a 0.35 M solution of ammonium chloride.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, arnold2619
What is the force of attraction between the particles in a salt crystal
Answers: 2

Chemistry, 22.06.2019 01:30, MickeyxX7096
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3


Chemistry, 22.06.2019 12:00, BreBreDoeCCx
Ageochemist examines a piece of metal that he found in the soil. he performs tests to identify the metal from its density, electrical conductivity, and melting point. which statement best describes his investigation? a. he is determining physical properties that are sufficient to identify the metal. b. he is determining chemical properties that are sufficient to identify the metal. c. he is determining physical properties that are insufficient to identify the metal. d. he is determining chemical properties that are insufficient to identify the metal.
Answers: 3
Do you know the correct answer?
Determine the mass of ammonium chloride, NH4Cl, required to prepare 0.250 L of a 0.35 M solution of...
Questions in other subjects:

Mathematics, 10.07.2019 08:00





Biology, 10.07.2019 08:00

History, 10.07.2019 08:00

Mathematics, 10.07.2019 08:00

Physics, 10.07.2019 08:00






