Chemistry, 13.04.2021 02:30, evelyngarcia99
Aqueous hydrochloric acid will react with solid sodium hydroxide to produce aqueous sodium chloride and liquid water . Suppose 1.5 g of hydrochloric acid is mixed with 2.67 g of sodium hydroxide. Calculate the maximum mass of water that could be produced by the chemical reaction. Round your answer to significant digits.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:00, tchase0616
What is the molarity of 60.0 grams of naoh dissolved in 750 milliliters of water? a) 1.1 m b) 2.0 m c) 12 m d) 75 m
Answers: 1

Chemistry, 22.06.2019 12:30, meghan2529
The melting point of sulfur is 115 °c and its boiling point is 445 °c. what state would sulfur be in at 200 °c?
Answers: 1
Do you know the correct answer?
Aqueous hydrochloric acid will react with solid sodium hydroxide to produce aqueous sodium chloride...
Questions in other subjects:




Mathematics, 30.08.2021 22:20

Mathematics, 30.08.2021 22:20


Mathematics, 30.08.2021 22:20




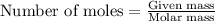 .....(1)
.....(1) 
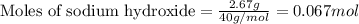

 of NaOH
of NaOH
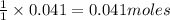 of water
of water