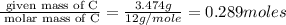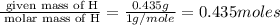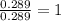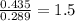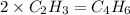Chemistry, 13.04.2021 02:00, staz13wiggins
When 3.915 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 12.74 grams of CO2 and 3.913 grams of H2O were produced. In a separate experiment, the molar mass of the compound was found to be 54.09 g/mol. Determine the empirical formula and the molecular formula of the hydrocarbon.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:30, alaynagrace1111
What is the mass of each element in a 324.8 sample of co2
Answers: 1

Chemistry, 22.06.2019 06:00, palomaresmitchelle
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1
Do you know the correct answer?
When 3.915 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 12.74 grams...
Questions in other subjects:

Mathematics, 24.02.2021 19:30

Mathematics, 24.02.2021 19:30

Chemistry, 24.02.2021 19:30

Mathematics, 24.02.2021 19:30

History, 24.02.2021 19:30


English, 24.02.2021 19:30

Health, 24.02.2021 19:30

Mathematics, 24.02.2021 19:30


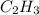 and
and  respectivley.
respectivley.
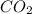 = 12.74 g
= 12.74 g
 = 3.913 g
= 3.913 g
 of carbon will be contained.
of carbon will be contained.
 of hydrogen will be contained.
of hydrogen will be contained.