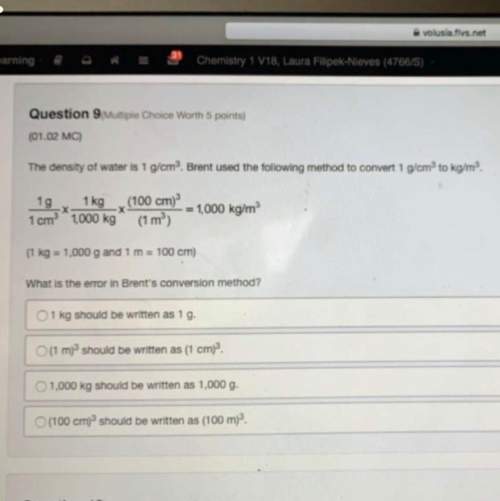
Ammonia (NH3 (g), Delta. Hf = –45.9 kJ/mol ) reacts with oxygen to produce nitrogen and water (H2 O(g), Delta. Hf = –241.8 kJ/mol) according to the equation below. 4 upper N upper H subscript 3 (g) plus 3 upper O subscript 2 (g) right arrow 2 upper N subscript 2 (g) plus 6 upper H subscript 2 upper O (g). What is the enthalpy of the reaction? Use Delta H r x n equals the sum of delta H f of all the products minus the sum of delta H f of all the reactants.. –3,572.1 kJ –1,267.2 kJ 1,267.2 kJ 3,572.1 kJ

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, officialrogerfp3gf2s
How to solve 4 nh3(g) + 5 o2(g) > 4 no(g) + 6 h2o(g) in chemistry
Answers: 1

Chemistry, 22.06.2019 15:00, Zagorodniypolina5
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 2

Chemistry, 22.06.2019 16:00, anaalashay
How will the volume of a gas be affected if the pressure is tripled, but the temperature remains the same?
Answers: 3

Chemistry, 22.06.2019 17:30, tiffanyhmptn
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1
Do you know the correct answer?
Ammonia (NH3 (g), Delta. Hf = –45.9 kJ/mol ) reacts with oxygen to produce nitrogen and water (H2 O(...
Questions in other subjects:

Mathematics, 05.06.2021 14:00

Mathematics, 05.06.2021 14:00

Mathematics, 05.06.2021 14:00

Mathematics, 05.06.2021 14:00

English, 05.06.2021 14:00

Mathematics, 05.06.2021 14:00

Mathematics, 05.06.2021 14:00

World Languages, 05.06.2021 14:00


Biology, 05.06.2021 14:00