

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, Falconpride4079
Construct the hypothetical phase diagram for metals a and b between room temperature (20c) and 700c, given the following information: * the melting temperature of metal a is 480c. • the maximum solubility of b in a is 4 wt% b, which occurs at 420c. • the solubility of b in a at room temperature is 0 wt% b. • one eutectic occurs at 420c and 18 wt% b–82 wt% a. • a second eutectic occurs at 475c and 42 wt% b–58 wt% a. • the intermetallic compound ab exists at a composition of 30 wt% b–70 wt% a, and melts congruently at 525c.• the melting temperature of metal b is 600c. • the maximum solubility of a in b is 13 wt% a, which occurs at 475c. • the solubility of a in b at room temperature is 3 wt% a.
Answers: 1

Chemistry, 22.06.2019 05:30, tifftiff22
Arecipe calls for 1.2 cups of oil. how many liters of oil is this?
Answers: 2

Do you know the correct answer?
A container at equilibrium contains the following concentrations: [Hz] - 0.30 M and [HI]
0.90 M. Th...
Questions in other subjects:

Mathematics, 04.10.2019 17:50

Business, 04.10.2019 17:50


English, 04.10.2019 17:50

English, 04.10.2019 17:50




Mathematics, 04.10.2019 17:50

Mathematics, 04.10.2019 17:50


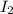 at equilibrium is 0.45 M
at equilibrium is 0.45 M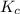
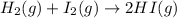
![K_c=\frac{[HI]^2}{[H_2]^1[I_2]^1}](/tpl/images/1245/8126/da9c3.png)
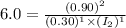
![[I_2]=0.45M](/tpl/images/1245/8126/936c9.png)




