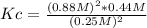Chemistry, 08.04.2021 09:40, itzdulceee
Calculate the value of Key in the following reaction if the equilibrium concentrations are
[H2S] -0.25 M, (H2) - 0.88 M, [S2] -0.44M.
2 H2Sig) = 2 H2(g) + S218)

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 07:30, bbyniah123
11. phosphorus-32 is radioactive and has a half life of 14 days. how much of a 124 mg sample of phosphorus-32 is present after 56 days? a) 7.75 mg b) 15.5 mg c) 31.0 mg d) 62.0 mg
Answers: 3

Chemistry, 22.06.2019 19:00, Farhan54019
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1
Do you know the correct answer?
Calculate the value of Key in the following reaction if the equilibrium concentrations are
[H2S] -0...
Questions in other subjects:



Mathematics, 05.11.2020 18:40

Mathematics, 05.11.2020 18:40

History, 05.11.2020 18:40





History, 05.11.2020 18:40


![Kc=\frac{[C]^{c} *[D]^{d} }{[A]^{a} *[B]^{b} }](/tpl/images/1245/8124/eda24.png)
![Kc=\frac{[H_{2} ]^{2} *[S_{2} ] }{[H_{2} S]^{2} }](/tpl/images/1245/8124/27cd3.png)