
Chemistry, 08.04.2021 08:30, sharkboy3780
A 10.0 mL sample of H2SO4 was exactly neutralized by 23.5 mL of 1.00 M KOH. Calculate the molarity of the acid. H2SO4(aq) + KOH(aq) —> K2SO4(aq) + H2O(l)

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 00:30, quintink
How can you write e method for the experiment of separating sand from water by filtration process? 1-materials 2-steps 3-conclusion also the same for the separating process of water and salt by filtration or distillation. quick because i need to finish my hw
Answers: 2
Do you know the correct answer?
A 10.0 mL sample of H2SO4 was exactly neutralized by 23.5 mL of 1.00 M KOH. Calculate the molarity o...
Questions in other subjects:





Mathematics, 07.04.2020 00:10


Social Studies, 07.04.2020 00:11




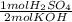 = 11.75 mmol H₂SO₄
= 11.75 mmol H₂SO₄



