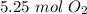Consider the following balanced equation:
2KCIO3(s) → 2KCl(s) + 302(g)
How many moles of O2 w...

Chemistry, 06.04.2021 23:20, hncriciacheichi
Consider the following balanced equation:
2KCIO3(s) → 2KCl(s) + 302(g)
How many moles of O2 will be obtained by decomposing
3.50 moles of KCIO3?
0.530 mole
O 3.00 moles
O 2.30 moles
5.25 moles

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 19:10, asdfghhk9805
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2

Chemistry, 22.06.2019 20:30, ashley4329
Select all the correct answers. which compounds have the empirical formula ch20? (multiple answers)a. c2h4o2b. c3h603c. ch2o2d. c5h1005e. c6h1206
Answers: 2
Do you know the correct answer?
Questions in other subjects:

Spanish, 17.01.2020 05:31




Mathematics, 17.01.2020 05:31

Chemistry, 17.01.2020 05:31



Mathematics, 17.01.2020 05:31

Mathematics, 17.01.2020 05:31


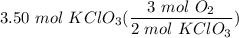 [DA} Multiply [Cancel out units]:
[DA} Multiply [Cancel out units]: