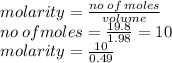Given this image, please try to decipher what the question is asking and work it out.
Given:
...

Chemistry, 06.04.2021 20:20, labrandonanderson00
Given this image, please try to decipher what the question is asking and work it out.
Given:
V = .49 L
m = 1.98 g/per packet
total mass = 19.8 g
I think that I am supposed to solve for molarity, but I don't think that there is enough information given. Thanks is advance & no fake answers please.


Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:30, ashleyjaslin
Calculate the expected ph values of the buffer systems from the experiments (a, b,c, d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 01:30, MickeyxX7096
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3


Chemistry, 22.06.2019 12:00, ctyrector
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1
Do you know the correct answer?
Questions in other subjects:

English, 03.09.2021 01:00



Mathematics, 03.09.2021 01:00