Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:20, UsedForSchool2018
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3



Chemistry, 22.06.2019 14:30, isaiahrodriguezsm17
What type(s) of intermolecular forces are expected between ch3ch2cooh molecules? dipole forces, induced dipole forces, hydrogen bonding
Answers: 1
Do you know the correct answer?
How many grams of Al2O3 can be formed when 3.33 moles of Al and 4.87 moles of O2 react according to...
Questions in other subjects:




Mathematics, 23.04.2021 17:10

Mathematics, 23.04.2021 17:10





Mathematics, 23.04.2021 17:10


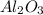
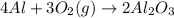
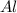 require = 3 moles of
require = 3 moles of 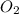
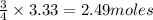 of
of 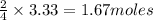 of
of