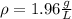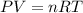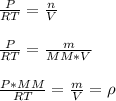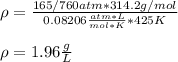Wally fluoride is an imaginary gaseous
compound with a molar mass of 314.2 g/mol.
(a) What is...

Chemistry, 02.04.2021 04:00, dbuchlerdb5599
Wally fluoride is an imaginary gaseous
compound with a molar mass of 314.2 g/mol.
(a) What is the density of wollmanium fluoride at 425 K
and 165 torr?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:40, yfgkeyonna
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 17:30, shookiegriffin
I'm learning about the periodic tables and what each subject's configuration is. for example, hydrogen is 1s^1, but i don't understand how you get that. can someone me understand how to figure out how to figure this out? sorry if the question makes no sense, but it would really a lot if you could me understand! you so much if you can!
Answers: 1
Do you know the correct answer?
Questions in other subjects:


Mathematics, 15.10.2020 04:01





English, 15.10.2020 04:01

Spanish, 15.10.2020 04:01


History, 15.10.2020 04:01