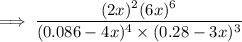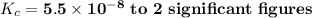Chemistry, 02.04.2021 01:40, shyanne5276
g Ammonia has been studied as an alternative "clean" fuel for internal combustion engines, since its reaction with oxygen produces only nitrogen and water vapor, and in the liquid form it is easily transported. An industrial chemist studying this reaction fills a tank with of ammonia gas and of oxygen gas, and when the mixture has come to equilibrium measures the amount of water vapor to be . Calculate the concentration equilibrium constant for the combustion of ammonia at the final temperature of the mixture. Round your answer to significant digits.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:10, bartonamber4042
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1

Chemistry, 22.06.2019 21:50, namoralessimon03
28. which is not a reason that water is used to store spent fuel rods from nuclear power plants? water increases the speed of the chain reaction in the fuel rods. water protects nuclear power plant workers from the high temperature and radiation of the fuel rods. water acts as a radiation shield to reduce the radiation levels. water cools the spent rods. salts action
Answers: 1

Chemistry, 23.06.2019 01:20, michellectucker1982
Use the de broglie's wave equation to find the wavelength of an electron moving at 7.3 × 106 m/s. show your work. note: h = plank's constant (6.62607 x 10-34 j s)
Answers: 1
Do you know the correct answer?
g Ammonia has been studied as an alternative "clean" fuel for internal combustion engines, since its...
Questions in other subjects:

Biology, 24.09.2019 04:30

Computers and Technology, 24.09.2019 04:30

Social Studies, 24.09.2019 04:30

Biology, 24.09.2019 04:30




Mathematics, 24.09.2019 04:30

Mathematics, 24.09.2019 04:30



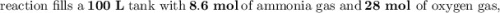
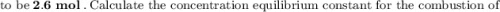
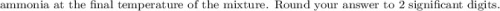
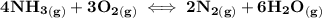
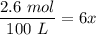
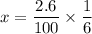
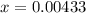
![\text{equilibrium constant} ({k_c}) = \dfrac{ [N_2]^2 [H_2O]^6 }{ [[NH_3]^4] [O_2]^3 }](/tpl/images/1236/3766/e00e9.png)