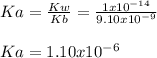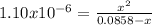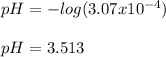Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:40, arlabbe0606
Effect of rotenone and antimycin a on electron transfer rotenone, a toxic natural product from plants, strongly inhibits nadh dehydrogenase of insect and fish mitochondria. antimycin a, a toxic antibiotic, strongly inhibits the oxidation of ubiquinol. (a) explain why rotenone ingestion is lethal to some insect and fish species. (b) explain why antimycin a is a poison. (c) given that rotenone and antimycin a are equally effective in blocking their respective sites in the electron-transfer chain, which would be a more potent poison? explain.
Answers: 3


Chemistry, 22.06.2019 19:20, evansh78
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3

Chemistry, 23.06.2019 01:30, kenldykido2300
Adirect relationship can be represented by: a curve a pie chart
Answers: 2
Do you know the correct answer?
Calculate the pH at the equivalence point when 22.0 mL of 0.200 M hydroxylamine, HONH2, is titrated...
Questions in other subjects:




Mathematics, 28.10.2020 18:00



Mathematics, 28.10.2020 18:00

English, 28.10.2020 18:00

English, 28.10.2020 18:00

Mathematics, 28.10.2020 18:00


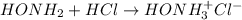

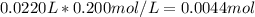
![[HONH_3^+Cl^-]=\frac{0.0044mol}{0.022L+0.0293L} =0.0858M](/tpl/images/1235/6200/36779.png)

![Ka=\frac{[H_3O^+][HONH_2]}{[HONH_3^+]}](/tpl/images/1235/6200/cfc73.png)