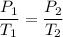Chemistry, 01.04.2021 07:00, sierrawalk3896
The pressure in a tire is 101 kPa at 10.0°C, what will be the pressure of a tire at 45.0°C?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 17:50, mytymikey123
You exhale co2 which is produced during cellular respiration. co2 combines with the water in your blood's plasma to make up one half of the body's most important buffer pair, carbonic acid. the more physical activity you engage in, the more co2 your body is producing. you can see this by putting some of the cabbage indicator in a glass and then blowing bubbles into it through a straw. can you see a change in the color of the indicator?
Answers: 2


Chemistry, 23.06.2019 15:30, Hunter1471
The gas in a sealed container has an absolute pressure of 9.25 atmospheres. if the air around the container is at standard pressure, what is the gauge pressure inside the container
Answers: 1

Chemistry, 23.06.2019 19:40, samariahmiddlebrooks
The reaction: (ch3)3cbr + oh− → (ch3)3coh + br− in a certain solvent is first order with respect to oh−. in several experiments, the rate constant k was determined at different temperatures. a plot of ln(k) vs 1/t was constructed and the slope of the line was -1.10 x 104 k with a y-intercept of 33.5. what is the value for k at a temperature of 25°c?
Answers: 1
Do you know the correct answer?
The pressure in a tire is 101 kPa at 10.0°C, what will be the pressure of a tire at 45.0°C?...
Questions in other subjects:

Mathematics, 25.08.2019 08:10





History, 25.08.2019 08:10

Mathematics, 25.08.2019 08:10