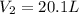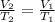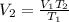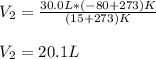Chemistry, 31.03.2021 08:10, molinaemily009
A balloon filled with helium has a volume of 30.0 L at a pressure of 100 kPa and a temperature of 15.0°C. What will the volume of the balloon be if the temperature is decreased to -80.0°C and the pressure remains constant?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 17:00, brownvester44
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2

Chemistry, 22.06.2019 22:30, eduardoguizar8787
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1

Chemistry, 23.06.2019 06:40, Science2019
How many joules of heat are required to raise thetemperature of 750 g of water from 11.0 °c to 19.0 °c?
Answers: 1
Do you know the correct answer?
A balloon filled with helium has a volume of 30.0 L at a pressure of 100 kPa and a temperature of 15...
Questions in other subjects:


Physics, 05.11.2021 22:30



Geography, 05.11.2021 22:30

Computers and Technology, 05.11.2021 22:30

Medicine, 05.11.2021 22:30

Business, 05.11.2021 22:30

Mathematics, 05.11.2021 22:30