
Chemistry, 29.03.2021 22:50, sonyalehardez
Look at the diagram of a voltaic cell below. Which half reaction occurs at the cathode in this cell?
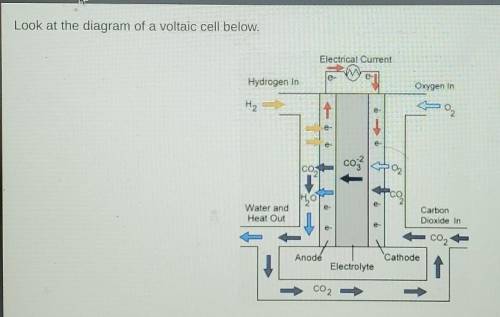

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, Gghbhgy4809
What pressure will be exerted by 0.675 moles of a gas at 25*c if it is in a 0.750-l container?
Answers: 1


Chemistry, 22.06.2019 14:30, hjlhdjfhjh
How does a noncompetitive inhibitor reduce an enzyme’s activity?
Answers: 1

Chemistry, 23.06.2019 10:30, EstherAbuwaah
Identify the limiting reactant when 9.65-g h2so4 reacts with 6.10-g of naoh. the equation is h2s04 + 2naoh = 2h2o + na2so4• what is the theoretical yield of na2so4, in grams? • how much of the excess reagent will remain after the reaction has been completed? • if 10.5-g of na2so4 are actually recovered experimentally, what is the percent yield?
Answers: 3
Do you know the correct answer?
Look at the diagram of a voltaic cell below.
Which half reaction occurs at the cathode in this cell...
Questions in other subjects:

English, 31.07.2019 04:00

History, 31.07.2019 04:00



Mathematics, 31.07.2019 04:00


Mathematics, 31.07.2019 04:00








