Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:40, taysomoneyyy
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Do you know the correct answer?
SOMEONE PLS HELP PLS PLS PLS
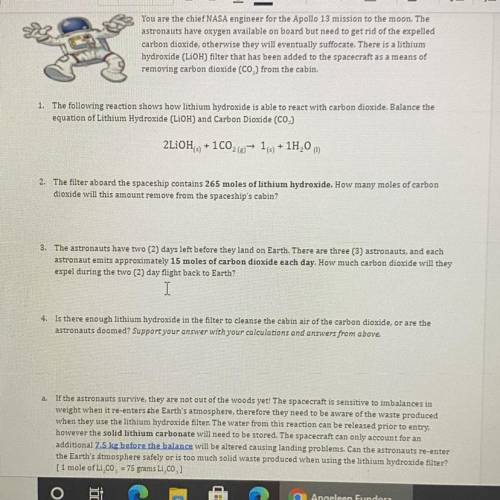
...
...
Questions in other subjects:

Mathematics, 26.03.2020 00:51


Arts, 26.03.2020 00:51





Mathematics, 26.03.2020 00:51

Chemistry, 26.03.2020 00:51