
Chemistry, 29.03.2021 16:50, lindamillscotton90
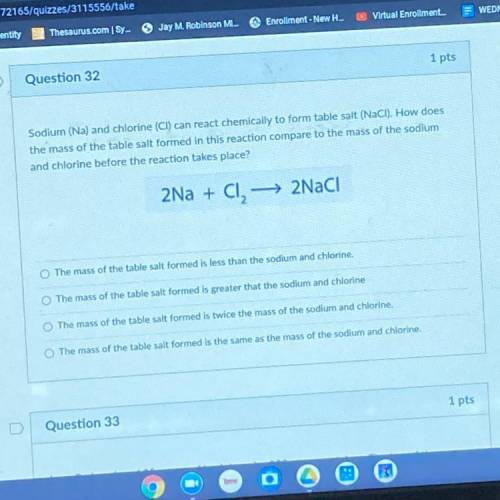
Sodium (Na) and chlorine (CI) can react chemically to form table salt (NaCl). How does the mass of the table salt formed in this reaction compare to the mass of the sodium and chlorine before the reaction takes place?

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 05:30, jalynholden07
Based on the formulas, select the compounds below that are covalent: kbr sif4 al2o3 co2 naco3 s7o2 pcl3 fe3n2 h2o s2f10
Answers: 3

Chemistry, 23.06.2019 08:00, oopsorry
Determine the number of moles of air present in 1.35 l at 750 torr and 17.0°c. which equation should you use? n=pv/rt what is the number of moles present? ⇒ 0.056 mol a sample of n2 gas occupying 800.0 ml at 20.0°c is chilled on ice to 0.00°c. if the pressure also drops from 1.50 atm to 1.20 atm, what is the final volume of the gas? which equation should you use? v2= p1v1t2/p2t1 what is the final volume of the gas? ⇒ 932 ml these are the answers
Answers: 1
Do you know the correct answer?
Sodium (Na) and chlorine (CI) can react chemically to form table salt (NaCl). How does
the mass of...
Questions in other subjects:


English, 23.02.2021 03:50



Business, 23.02.2021 03:50


Mathematics, 23.02.2021 03:50



Mathematics, 23.02.2021 03:50






