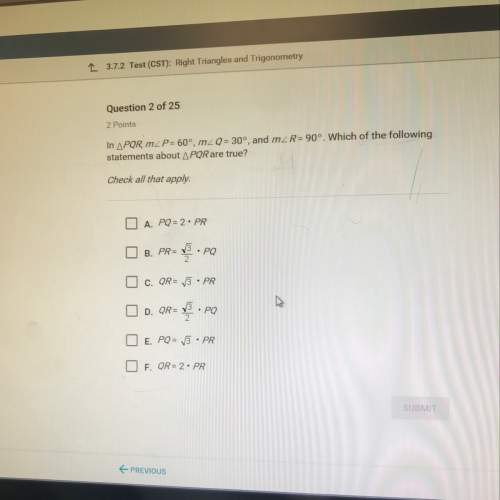
17. Consider the following reaction:
4Al(s) + 302(8) 2A1,03(s)
It takes 2.00 L of pure oxygen...

Chemistry, 28.03.2021 19:50, diamondalize21p84czi
17. Consider the following reaction:
4Al(s) + 302(8) 2A1,03(s)
It takes 2.00 L of pure oxygen gas at STP to react completely with a certain sample of
aluminum. What is the mass of aluminum reacted?
18.

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 00:30, cashkidd2200
There are approximately 15 milliliters (ml) in 1 tablespoon (tbsp). what expression can be used to find the approximate number of milliliters in 3 tbsp?
Answers: 1

Chemistry, 23.06.2019 05:00, lifeoflashay1659
He nucleus contains the cells genetic material in the form of dna. dna is organized into our chromosomes, which are made up of thousands of that determine our traits.
Answers: 1
Do you know the correct answer?
Questions in other subjects: