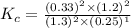Chemistry, 26.03.2021 21:30, ariellake8551
Nitrogen and water react to form nitrogen monoxide and hydrogen, like this: N2(g) + 2H2O(g) → 2NO(g) +2H2(g)Also, a chemist finds that at a certain temperature the equilibrium mixture of nitrogen, water, nitrogen monoxide, and hydrogen has the following composition: compound pressure at equilibrium N2 0.25 M H20 1.3 M NO 0.33 M H2 1.2 MCalculate the value of the equilibrium constant for this reaction. Round your answer to significant digits.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, skaterwolf1317
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2


Chemistry, 23.06.2019 00:00, ahmedeldyame
#20 which type of bond is formed when bases pair in dna? ionic bond covalent bond coordinate bond hydrogen bond
Answers: 1

Chemistry, 23.06.2019 01:00, aliviadushane
If a straight-chain hydrocarbon is a gas at room temperature, how many carbon atoms will it have? a. 6 carbon atoms b. 12 carbon atoms c. 24 carbon atoms d. 3 carbon atoms
Answers: 1
Do you know the correct answer?
Nitrogen and water react to form nitrogen monoxide and hydrogen, like this: N2(g) + 2H2O(g) → 2NO(g)...
Questions in other subjects:






German, 02.03.2021 03:00



Mathematics, 02.03.2021 03:00

Spanish, 02.03.2021 03:00


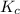
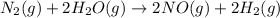
![K_c=\frac{[NO]^2\times [H_2]^2}{[H_2O]^2\times [N_2]^1}](/tpl/images/1224/3364/cca59.png)