
Chemistry, 25.03.2021 22:40, heleneallie7ou1fnk
The temperature of water decreases on the dissolution of NH4NO3. From this observation, we can conclude that the solution process would be best described asa)exothermic with a positive entropy change. b)exothermic with a negative entropy change. c)endothermic with a negative entropy change. d)endothermic with a positive entropy change. e)exothermic with a negative free energy change

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:40, justicejesusfreak
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1

Chemistry, 22.06.2019 19:50, VoidedAngel
When the mercury level in a barometer decreases that atmospheric pressure has
Answers: 3

Chemistry, 22.06.2019 21:40, k3rbycalilung
Tooth enamel consists mainly of the mineral calcium hydroxyapatite, ca_10(po_4)_6(oh)_2. trace elements in teeth of archaeological specimens provide anthropologist with clues about diet and diseases of ancient people. students at hamline university measured strontium in enamel from extracted wisdom teeth by atomic absorption spectroscopy. solutions with a constant total volume of 10.0 ml contained 0.726 mg of dissolved tooth enamel plus variable concentrations of added sr. added sr find the concentration of sr in the 10 ml sample solution in parts per billion = ng/ml. find the concentration of sr in tooth enamel in parts per million = mu g/g.
Answers: 2
Do you know the correct answer?
The temperature of water decreases on the dissolution of NH4NO3. From this observation, we can concl...
Questions in other subjects:

History, 18.10.2019 05:10



Chemistry, 18.10.2019 05:10

Mathematics, 18.10.2019 05:10

Mathematics, 18.10.2019 05:10


Mathematics, 18.10.2019 05:10

Social Studies, 18.10.2019 05:10

Mathematics, 18.10.2019 05:10


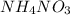 , it means heat has been absorbed by
, it means heat has been absorbed by 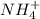 and
and 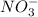 , randomness will increase and thus there is a positive entropy change.
, randomness will increase and thus there is a positive entropy change.



