These are the rest of the questions right down below
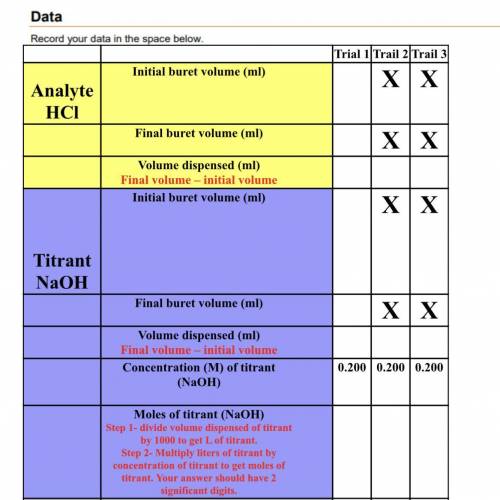
Analyte
HCl
Mole of Analyte (HCl)<...

Chemistry, 25.03.2021 18:00, diegorivas7992
These are the rest of the questions right down below
Analyte
HCl
Mole of Analyte (HCl)
(Equal to the moles of titrant)
Concentration (M)of analyte (HCl)
Step 1- divide volume dispensed of analyte by 1000 to get L of analyte
Step 2- Divide moles of analyte by liters of analyte to get concentration.
Average concentration(M) of analyte.
Add up the analyte concentrations from the three trials. Divide your answer by 3. Include 3 significant digits in your answer.
Percent error of concentration (M) of analyte.
Actual concentration of HCl = 0.120 M
Experimental concentration- Use the average you calculated.
Step 1- Subtract experimental value from actual value.
Step 2- Divide answer in Step 1 by actual value.
Step 3- Multiply answer in Step 3 by 100.
Your answer should be expressed as a percentage.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:50, mrylenastewart
What are transitions between a liquid and a solid called? identify which way they are transitioning
Answers: 2

Do you know the correct answer?
Questions in other subjects:



Mathematics, 12.06.2021 03:50

Mathematics, 12.06.2021 03:50



Mathematics, 12.06.2021 03:50

Business, 12.06.2021 03:50

Health, 12.06.2021 03:50







