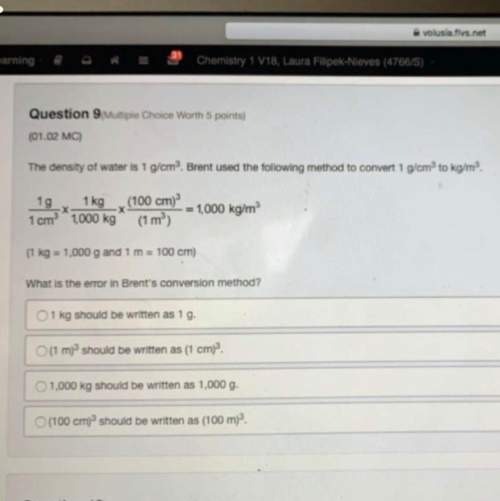
We have seen that adding the common ion (salt of the conjugate base) to a weak acid equilibrium changes the pH. Imagine a solution in which you have created a solution that is 2.0 M in acetic acid and 2.0 M in sodium acetate (the purple atom is the sodium cation). In this case the concentration of the weak acid and it's conjugate base are exactly equal. In truth, a small amount of acetate anion (conjugate base) is also coming from the dissociation of the weak acid, but this is negligible compared to the added sodium acetate.
a. The Na+ cation does not appear in the equilibrium because it is a spectator ion and is only the counter ion for the acetate anion.
b. All of the acetic acid will become deprotonated by water so the final solution contains ony aqueous h aron un ions and acetate ons.
c. The pKa for the acid = the pH of this solution. Since acetic acid is a weak acid, acetate anion is considered to be a strong base.
d. The [H3O+]=Ka in this solution. The pH of this solution will be lower than that of a 2.0 M solution of acetic acid.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, gilbert325
Write the chemical symbols for three different atoms or atomic cations with 27 electrons. asap!
Answers: 2

Chemistry, 23.06.2019 02:30, hailee232
When the ionic compound nabr dissolves in water, br– ions are pulled into solution by the attraction between what two particles? a. the na+ and br– ions b. the na+ ion and the negative end of a water molecule c. the br– ion and the positive end of a water molecule d. the br– ion and the negative end of a water molecule
Answers: 1

Chemistry, 23.06.2019 06:00, hopechinn6646
Complete the sentences to best explain the ranking. match the words below to the appropriate blanks in the sentences. a less polar bondhigher molar massion-dipole forcesstronger intermolecular forcesdipole-dipole forcesdispersion forceshydrogen bonding1. h2s and h2se exhibit the following intermolecular forces:.2. therefore, when comparing h2s and h2se the one with a has a higher boiling point .3. the strongest intermolecular force exhibited by h2o is . therefore, when comparing h2se and h2o the one with has a higher boiling point.
Answers: 1

Chemistry, 23.06.2019 10:20, Thejollyhellhound20
Based on the equation, how many grams of br2 are required to react completely with 29.2 grams of alcl3? alcl3 + br2 → albr3 + cl2 48.7 grams 52.6 grams 56.7 grams 61.3 grams
Answers: 3
Do you know the correct answer?
We have seen that adding the common ion (salt of the conjugate base) to a weak acid equilibrium chan...
Questions in other subjects:

History, 05.05.2020 13:29


History, 05.05.2020 13:29



Mathematics, 05.05.2020 13:29



Mathematics, 05.05.2020 13:29