
Chemistry, 25.03.2021 16:30, kirkhester1
1 mole of oxygen gas and 2 moles of hydrogen are mixed in a vessel at a constant temperature, and no chemical reaction occurs. If the total pressure inside the vessel is 3 atm, what is the partial pressure of the hydrogen gas?
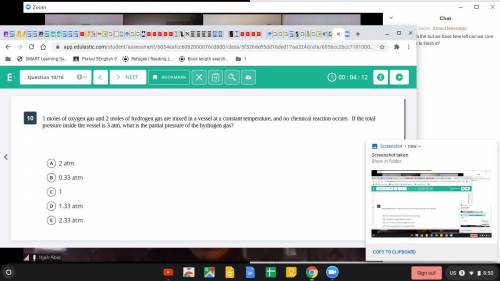

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 03:30, ruleolivas
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 05:30, sethjohnson386pbnm3x
Modern weaponry has increased the number of deaths in wars and violent conflicts.
Answers: 3

Chemistry, 22.06.2019 10:10, andersonemma2222
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1
Do you know the correct answer?
1 mole of oxygen gas and 2 moles of hydrogen are mixed in a vessel at a constant temperature, and no...
Questions in other subjects:

History, 16.10.2019 21:10

English, 16.10.2019 21:10



History, 16.10.2019 21:10




Mathematics, 16.10.2019 21:10






