
Chemistry, 25.03.2021 05:10, gabriel5575
PLEASE HELP
1. A gas with a volume of 4.0 L at a pressure of 2.03 atm is allowed to expand to a volume of 12.0 L. What is the pressure in atm in the container if the temperature remains constant. Please show work (including the equation you used).
2.The volume of a gas is 200 mL at 350.0 kPa pressure. What will the volume be when the pressure is reduced to 120.0 kPa, assuming the temperature remains constant. Please show all work for full credit. Please show work (including the equation you used).
3.What is the original pressure of a bike tire if the current pressure is 150 atm and the volume changed from 1000 mL to 700 mL. Please show work (including the equation you used).

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 13:30, kkingstone7062
What does the xylem do? stores the glucose captures the sunlight absorbs oxygen into the leaf carries water from the roots to the leaves
Answers: 1

Do you know the correct answer?
PLEASE HELP
1. A gas with a volume of 4.0 L at a pressure of 2.03 atm is allowed to expand to a vol...
Questions in other subjects:








Mathematics, 13.10.2020 04:01


Mathematics, 13.10.2020 04:01


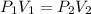 (also called Boyle's Law), where P₁ and V₁ denote the initial pressure and volume, and P₂ and V₂ denote the final (or changed) pressure and volume, respectively.
(also called Boyle's Law), where P₁ and V₁ denote the initial pressure and volume, and P₂ and V₂ denote the final (or changed) pressure and volume, respectively.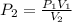 .
. ![\[P_2=\frac{\left ( 2.03 \text{ atm} \right )\left ( 4.0 \text{ L} \right )}{\left ( 12.0 \text{ L} \right )}\] = 0.67 atm.](/tpl/images/1219/2359/100f6.png)
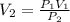
![\[V_2=\frac{\left ( 350.0 \text{ kPa} \right )\left ( 200 \text{ mL} \right )}{\left ( 120.0 \text{ kPa} \right )}\] = 583 mL.](/tpl/images/1219/2359/ee8b8.png)
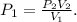
![\[P_1=\frac{\left ( 150 \text{ atm} \right )\left ( 700 \text{ mL} \right )}{\left ( 1000 \text{ mL} \right )}\] = 105 atm.](/tpl/images/1219/2359/a3484.png)




