
Chemistry, 25.03.2021 03:20, galaxicorn45
I've been stuck on this
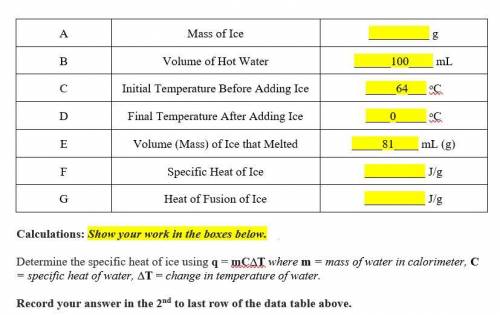
Determine the specific heat of ice using q = mC∆T where m = mass of water in calorimeter, C = specific heat of water, ∆T = change in temperature of water.
Determine the heat of fusion of ice using q = m∆Hf where q = heat, m = mass of substance being melted, ∆Hf = heat of fusion of substance being melted.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:10, akatsionis25
When will le chatelier's principle come into effect? at the beginning of a reaction, when there are only reactants when a reaction has reached chemical equilibrium when a catalyst is added to a reaction mixture when a reaction is occurring but not yet at equilibrium
Answers: 3

Chemistry, 22.06.2019 01:30, MickeyxX7096
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3


Chemistry, 22.06.2019 22:30, kiera2599
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1
Do you know the correct answer?
I've been stuck on this
Determine the specific heat of ice using q = mC∆T where m = mass of water i...
Questions in other subjects:

Mathematics, 19.02.2021 04:30



Mathematics, 19.02.2021 04:30


Mathematics, 19.02.2021 04:30


Mathematics, 19.02.2021 04:30







