
Chemistry, 24.03.2021 20:30, franklinkaylieg3873
Two possible Lewis structures for the molecule HCN are given. Determine the formal charge on each atom in both structures.
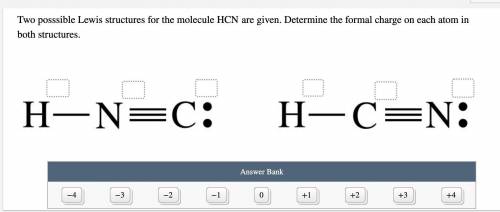

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, gwenparks
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1


Chemistry, 22.06.2019 23:30, jade468
Substance a is a nonpolar liquid and has only dispersion forces among its constituent particles. substance b is also a nonpolar liquid and has about the same magnitude of dispersion forces among its constituent particles. when substance a and b are combined, they spontaneously mix.
Answers: 1
Do you know the correct answer?
Two possible Lewis structures for the molecule HCN are given. Determine the formal charge on each at...
Questions in other subjects:

Mathematics, 08.07.2019 19:30

Mathematics, 08.07.2019 19:30

Chemistry, 08.07.2019 19:30




French, 08.07.2019 19:30

Mathematics, 08.07.2019 19:30







