If you dissolve 40g of KCI at 80c, the solution would be?
unsaturated or super saturated...

Chemistry, 24.03.2021 08:00, cakeisalie6865
If you dissolve 40g of KCI at 80c, the solution would be?
unsaturated or super saturated
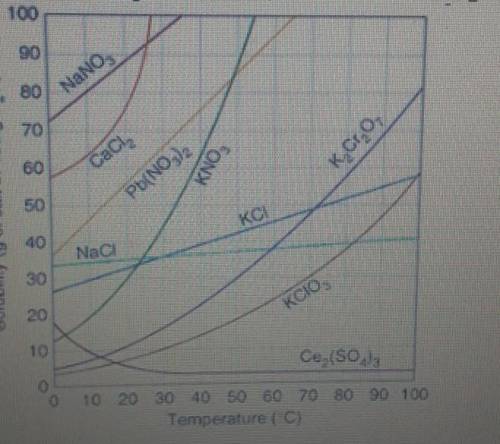

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 20:10, jakhunter354
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 23.06.2019 01:10, dontcareanyonemo
Can someone check my work 98 5.05 acids and bases for this assignment you will be comparing acids and bases. the chart below will you organize the information needed: acids bases chemical properties (2) deodorant detergent vinger dish soap physical properties (2) orange juice toilet cleaner drain cleaner window cleaner ph level acid ph goes from 0-4 bases ph goes from 10-14 examples around you (2) vinger coffee lemon juice dark chocolate
Answers: 3


Chemistry, 23.06.2019 15:00, ericperkins10ox0b27
In two or more complete sentences describe all of the van der waals forces that exist between molecules of sulfur dioxide, so2.
Answers: 1
Do you know the correct answer?
Questions in other subjects:


Chemistry, 13.11.2020 01:00

Social Studies, 13.11.2020 01:00



Health, 13.11.2020 01:00

Mathematics, 13.11.2020 01:00


English, 13.11.2020 01:00

Mathematics, 13.11.2020 01:00






