
Chemistry, 05.11.2019 01:31, Kinglilray998
Acompound is 2% h, 32.7% s, and 65.3% o by mass. what is the subscript on the o in the empirical formula for this compound?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:00, hdjsjfjruejchhehd
Define dew point. i am writing this part to be able to ask the question
Answers: 1


Chemistry, 22.06.2019 20:20, catchonyet
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1
Do you know the correct answer?
Acompound is 2% h, 32.7% s, and 65.3% o by mass. what is the subscript on the o in the empirical for...
Questions in other subjects:


History, 17.12.2019 20:31







Chemistry, 17.12.2019 20:31


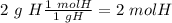
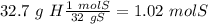
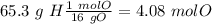
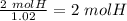
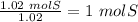
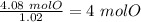
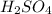 with a "4" for oxygen.
with a "4" for oxygen.




