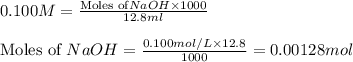Chemistry, 22.03.2021 22:30, jayden6467
molecular formula citric acid2.A 10.0 mL sample of pineapple juice was titrated with 0.100 M sodium hydroxide solution. The average volume of NaOH required to reach the endpoint was 12.8 mL. a. Calculate the number of moles of sodium hydroxide required to reach the endpoint. Show your work in equation editor. Remember to use units and report your answer to the proper number of significant figures.

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 00:10, graceception
In as 1°, 2°, 3°, or 4°. be to . : °b: °c: °d: ° : °b: °c: °d: ° : °b: °c: °d: °e: °f: °g: °h: ° : °b: °c: °d: °e: °f: °g: °h: °i: °
Answers: 3

Chemistry, 23.06.2019 00:30, Keemdadream13
If there are 3.5 moles of koh, how many moles of naoh can be produced? question 1 options: a)3.0 moles naoh b)3.5 moles naoh c)1 moles naoh d)9 moles naoh
Answers: 1
Do you know the correct answer?
molecular formula citric acid2.A 10.0 mL sample of pineapple juice was titrated with 0.100 M sodium...
Questions in other subjects:






English, 03.12.2019 07:31

Mathematics, 03.12.2019 07:31



History, 03.12.2019 07:31


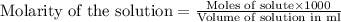
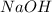 solution = 0.100 M
solution = 0.100 M