
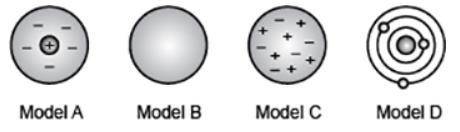
The diagram shows the different models of the atom that eventually led to the modern atomic theory. Four diagrams are shown labeled Model A, Model B, Model C, Model D from left to right. The diagram labeled Model A has a small sphere with a positive sign on it. This small sphere at the center is surrounded by a shaded circle. The shaded circle has negative signs placed randomly inside it. The diagram labeled Model B is a large shaded solid sphere. The diagram labeled Model C has positive and negative signs scattered in a shaded sphere. The diagram labeled Model D has a shaded sphere surrounded by three concentric circles. Each concentric circle has a sphere on it. Which model was proposed as a result of Rutherford's scattering experiment where positive particles did not pass straight through a foil as expected? (5 points) Model A Model B Model C Model D

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 04:40, twinchristiansp4xhd2
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1

Chemistry, 23.06.2019 06:00, lanaiheart7
What are the coefficients to balance the following equation? ba+br2=babr2
Answers: 2

Chemistry, 23.06.2019 10:00, lexusdixon3
Which number should be placed before f2 on the reactants side equation to make equation balanced? xe + > xef4
Answers: 1
Do you know the correct answer?
The diagram shows the different models of the atom that eventually led to the modern atomic theory....
Questions in other subjects:




Mathematics, 30.05.2020 15:59

History, 30.05.2020 15:59










