
Chemistry, 21.03.2021 06:50, samiller30
A volume of 10cm3 of sulfuric acid of unknown concentration neutralized 25cm3 of sodium hydroxide solution of concentration 0.4 mol dm-3. The concentration of the acid is:
0.1 mol dm-3
0.25 mol dm-3
0.4 mol dm-3
0.5 mol dm-3

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 10:10, babyphoraaaaa
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate, m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 22.06.2019 22:10, steven0448
Determine the ph of 0.10 m nh3 solution. nh3 is a weak base with a kb equal to 1.8 x 10-5 round answer to nearest whole number.
Answers: 1
Do you know the correct answer?
A volume of 10cm3 of sulfuric acid of unknown concentration neutralized 25cm3 of sodium hydroxide so...
Questions in other subjects:

Physics, 20.11.2019 00:31


Mathematics, 20.11.2019 00:31





Chemistry, 20.11.2019 00:31



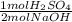 = 0.005 mol H₂SO₄
= 0.005 mol H₂SO₄



