
Chemistry, 21.03.2021 06:20, 1r32tgy5hk7
A 2.455 g sample of a new organic material is combusted in a bomb calorimeter. The temperature of the calorimeter and its contents increase from 23.61 ∘C to 27.04 ∘C. The heat capacity (calorimeter constant) of the calorimeter is 38.29 kJ/ ∘C, what is the heat of combustion per gram of the material?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, carsonjohnsonn
If a 12-v battery is connected to a circuit that has a current of 3.0 a, what is the total resistance in the circuit? 36 ohms 4 ohms 0.25 ohms
Answers: 1

Chemistry, 22.06.2019 07:30, kimberlyrios12p0ts98
Which of the following best supports the concept that genetic information is passed on to offspring from both of their parents, not just one?
Answers: 2

Do you know the correct answer?
A 2.455 g sample of a new organic material is combusted in a bomb calorimeter. The temperature of th...
Questions in other subjects:

Mathematics, 20.07.2019 20:00

History, 20.07.2019 20:00


Biology, 20.07.2019 20:00

History, 20.07.2019 20:00

Mathematics, 20.07.2019 20:00





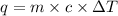
 = Heat gained by calorimeter
= Heat gained by calorimeter
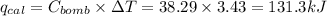
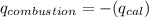
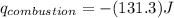
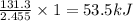 of heat
of heat




