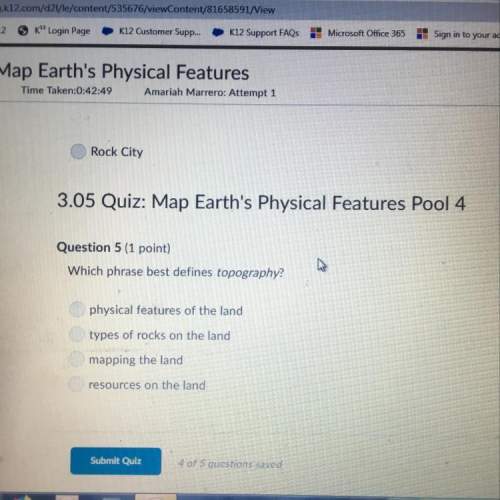
Chemistry, 21.03.2021 04:30, gwendallinesikes
Very long question but PLZ help
20. Calculate the hydrogen-ion concentration [H + ] for the aqueous solution in which [OH – ]
is 1 x 10 –11 mol/L. Is this solution acidic, basic, or neutral? Show your work. (3 points) Use
K w to solve for the hydrogen ion concentration here. Then, once you have your numerical
answer (with units!), answer the question about the solution.
Calculate the hydrogen-ion concentration (show all set up work with calculations) – (2 pts)
Discuss your answer and how it is related to the solution being acidic, basic or neutral and then
identify your solution as acidic, basic or neutral based on your calculated hydrogen-ion
concentration. – (1 pts)

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, Sarahinator04
0.09 moles of sodium sulfate in 12 ml of solution
Answers: 3



Chemistry, 22.06.2019 21:00, alwaysneedhelp84
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1
Do you know the correct answer?
Very long question but PLZ help
20. Calculate the hydrogen-ion concentration [H + ] for the aqueous...
Questions in other subjects:


Mathematics, 06.05.2020 08:10

Advanced Placement (AP), 06.05.2020 08:10

Biology, 06.05.2020 08:10


History, 06.05.2020 08:10


Mathematics, 06.05.2020 08:10