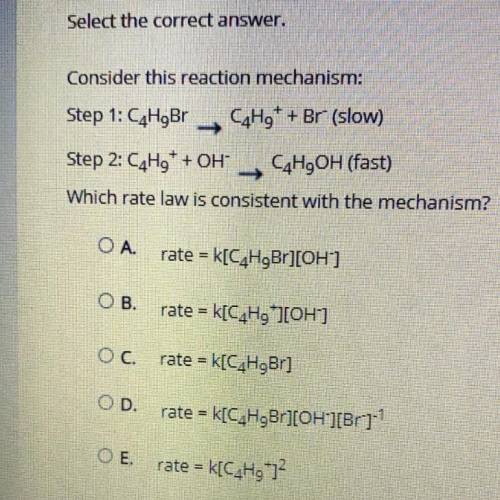
Consider this reaction mechanism:
Step 1: C4H9Br C4H9+ + Br- (slow)
Step 2: C4H9+ + OH- C4H9O...


Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:10, Kianna000
56.16 gregor mendel was the first scientist to use statistics to analyze scientific data. before mendel's experiments, scientists believed that organisms acquired traits from their environment and passed them on to their offspring. after mendel's discoveries were accepted, scientists realized that traits passed to offspring were the result of genes being passed from parents to offspring. this is an example of pls
Answers: 1

Chemistry, 22.06.2019 10:40, trinityanne1738
Asolid that forms and separates from a liquid mixture is called
Answers: 2

Chemistry, 22.06.2019 16:50, lilblackbird4
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 20:30, ashley4329
Select all the correct answers. which compounds have the empirical formula ch20? (multiple answers)a. c2h4o2b. c3h603c. ch2o2d. c5h1005e. c6h1206
Answers: 2
Do you know the correct answer?
Questions in other subjects:

Chemistry, 28.05.2021 14:00

Computers and Technology, 28.05.2021 14:00

Mathematics, 28.05.2021 14:00


Mathematics, 28.05.2021 14:00



English, 28.05.2021 14:00

Mathematics, 28.05.2021 14:00

Physics, 28.05.2021 14:00