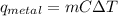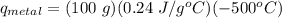Chemistry, 19.03.2021 21:50, momneedshelphmwk
A student investigates a pure metal, X . The student takes a 100.0 g piece of metal X , heats it to 500.0°C , then places it on a 1000.0 g block of ice at 0.0°C . The ice partially melts, and the final temperature of the metal, ice, and melted water is 0.0°C . The student calculates the experimental value of the specific heat capacity of metal X and records it as 0.24 J/(g⋅°C) . Calculate the magnitude of the energy change (qmetal) of metal X during the experiment.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 13:30, bryce99
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2


Chemistry, 22.06.2019 19:40, jholland03
What type of electromagnetic waves does the human eye see as the colors red blue or green a visible light waves b radio waves c infrared waves d microwaves
Answers: 1
Do you know the correct answer?
A student investigates a pure metal, X . The student takes a 100.0 g piece of metal X , heats it to...
Questions in other subjects:


Physics, 03.08.2021 14:00

Business, 03.08.2021 14:00

English, 03.08.2021 14:00

Mathematics, 03.08.2021 14:00

Mathematics, 03.08.2021 14:00


English, 03.08.2021 14:00

Mathematics, 03.08.2021 14:00

English, 03.08.2021 14:00