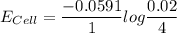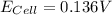Chemistry, 19.03.2021 18:20, sunshine52577oyeor9
A certain metal M forms a soluble nitrate salt M(NO3), Suppose the left half cell ofa galvanic cell apparatus is filled with a 4.00 M solution of M (NO,), and the right half cell with a 20.0 mM solution of the same substance. Electrodes made of M are dipped into both solutions and a voltmeter is connected between them. The temperature of the apparatus is held constant at 35.0 °C.
Required:
a. Which electrode will be positive?
b. What voltage will the voltmeter show?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:00, WhiteWinterRose
What is the chemical formula of the following compound
Answers: 3

Chemistry, 21.06.2019 18:00, Queenquestion5967
When the following equation is balanced using the smallest possible integers, what is the coefficent of oxygen gas? c7h16o(g) + o2(g) → co2(g) + h2o(g) -1 -5 -8 -16 -21
Answers: 3


Chemistry, 22.06.2019 07:30, 10040813
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3
Do you know the correct answer?
A certain metal M forms a soluble nitrate salt M(NO3), Suppose the left half cell ofa galvanic cell...
Questions in other subjects:





Mathematics, 30.04.2021 22:30

Physics, 30.04.2021 22:30




Mathematics, 30.04.2021 22:30