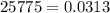Chemistry, 18.03.2021 03:10, hiiamsuperverylong
Methane gas is collected over water at 25 C and an atmospheric pressure of 775 mmHg. What is the dry pressure of the gas at this temperature? (Vapor Pressure of water = 0.0313 atm).

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, sotoamerica0814
98 ! and brainliest plz ! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1


Chemistry, 22.06.2019 19:30, simihehe
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2

Chemistry, 22.06.2019 20:30, sydneip6174
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2
Do you know the correct answer?
Methane gas is collected over water at 25 C and an atmospheric pressure of 775 mmHg. What is the dry...
Questions in other subjects:

Mathematics, 04.12.2020 18:40


Mathematics, 04.12.2020 18:40

Chemistry, 04.12.2020 18:40

Biology, 04.12.2020 18:40



Advanced Placement (AP), 04.12.2020 18:40


Mathematics, 04.12.2020 18:40