
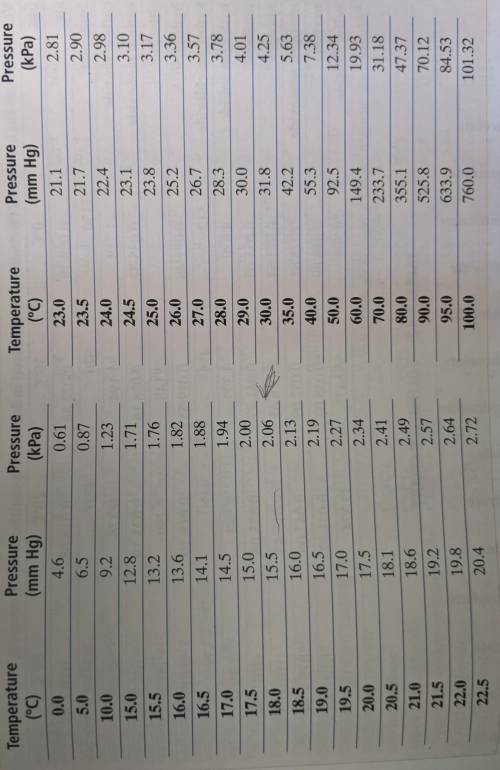
Can someone please help? This was due yesterday. The Appendix Table A-8 is linked in a photo. A certain mass of oxygen was collected over water when potassium chlorate was decomposed by heating. The volume of the oxygen sample collected was 720. mL at 25.0° C and a barometric pressure of 755 torr. What would the volume of the oxygen be at STP? (Hint: First calculate the partial pressure of the oxygen, using Appendix Table A-8. Then use the combined gas law.)

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:30, jameskarbar9p8c9d2
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1

Chemistry, 22.06.2019 07:10, jasondesatnick
An experimental procedure requires a 10 ml of acid to be dissolved
Answers: 2

Chemistry, 22.06.2019 19:30, simihehe
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2
Do you know the correct answer?
Can someone please help? This was due yesterday. The Appendix Table A-8 is linked in a photo.
A cer...
Questions in other subjects:

Mathematics, 09.07.2019 13:40


Mathematics, 09.07.2019 13:40



History, 09.07.2019 13:40

History, 09.07.2019 13:50

History, 09.07.2019 13:50

History, 09.07.2019 13:50

Social Studies, 09.07.2019 13:50






